Answers
Answer:
UK stop spending a lot on clothes if u don't wear half of them
Related Questions
Describe the trend of the reactivity of the elements in group VII
Answers
The non-metal elements in Group 7 – known as the halogens – get less reactive as you go down the group
Answer & Explanation:
The reactivity of elements in Group VII, also known as Group 17, decreases with increasing atomic radius. This is because halogens have high electronegativities and a proclivity to gain electrons in noble gas configurations. Myths are traditional stories or beliefs that explain cultural or societal beliefs, customs, or natural phenomena. They can be passed down through generations and can be based on true or fictitious events. Mythology, on the other hand, is the collection of myths associated with a specific culture or religion. Mythology can be amplified through retelling, incorporation into religious practices; association with significant events or figures, and adaptation into other media forms such as literature, film, or art.
The energy required for the chemical reaction of 21.7 grams of nitrogen to produce ammonia is how many kilojoules? If you put something that isn't the answer I will report you.
Answers
Answer:
\(Q=-71.1kJ\)
Explanation:
Hello!
In this case, since the formation of ammonia by starting with nitrogen and therefore hydrogen is:
\(N_2+3H_2\rightarrow 2NH_3\)
Which has an energy of reaction of:
\(\Delta _fH_{NH_3}=-45.90 \frac{kJ}{molNH_3}\)
We can compute the energy required for this reaction by first computing the moles of ammonia yielded by 21.7 grams of nitrogen (28.02 g/mol) via stoichiometry:
\(n_{NH_3}=21.7gN_2*\frac{1molN_2}{28.02gN_2}*\frac{2molNH_3}{1molN_2}=1.55molNH_3\)
Thus, the energy turns out:
\(Q=n_{NH_3}\Delta _fH_{NH_3}=1.55molNH_3 * -45.90 \frac{kJ}{molNH_3}\\\\Q=-71.1kJ\)
Best regards!
Answer: 71.1 (Edmentum)
Explanation:
Which of the following describes the Aufbau principle?
a) each electron is added to the lowest energy level orbital available
b) none of the above
c) the lowest energy configuration for an atom is the one with maximum number of unpaired electrons
d) no 2 electrons in the same atom can have the same set of 4 quantum numbers.
Answers
The statement that describes the Aufbau principle is ; ( A ) each electron is added to the lowest energy level orbital available
The Aufbau principle states that at ground level of an atom/ion the orbitals of the lowest energy levels of the atom are first filled up by electrons. therefore
It can be said that each electron is added to the lowest energy level orbital available before electrons will be added to orbitals with higher energy levels.
Hence we can conclude that The statement that describes the Aufbau principle is each electron is added to the lowest energy level orbital available
Learn more : https://brainly.com/question/13393709

Group A elements are considered representative elements. True False
Answers
Answer:
This is True
Explanation:
If you look at the elemetnts and use notes, you can see it is a represetaive element. have a nice day! good luck!
NO + H2-> N2 + H2O
Balance equation?
Answers
Answer:
2NO + 4H-> N2 + 2 H2O
Explanation:
Both sides must be equal. :)
How would the number 3475.32 in be expressed in scientific notation? *
Answers
Explanation:
i think 3x10^5
hope this helps if not sorry
1) What two types of substances create ionic compounds?
2) What substances create molecular compounds?
NEED HELP ASAP PLEASE!
Answers
Answer:
1. Ionic compounds generally form between elements that are metals and elements that are nonmetals. For example, the metal calcium (Ca) and the nonmetal chlorine (Cl) form the ionic compound calcium chloride (CaCl2).
2.A binary molecular compound is a molecular compound that is composed of two elements. In general, the elements that combine to form binary molecular compounds are both nonmetals. This contrasts with ionic compounds, which usually involve bonds between metal ions and nonmetal ions
Answer:
1. Ionic compounds generally form between elements that are metals and elements that are nonmetals. For example, the metal calcium (Ca) and the nonmetal chlorine (Cl) form the ionic compound calcium chloride (CaCl2).
2.A binary molecular compound is a molecular compound that is composed of two elements. In general, the elements that combine to form binary molecular compounds are both nonmetals. This contrasts with ionic compounds, which usually involve bonds between metal ions and nonmetal ions
Explanation:
Choose the option that would convert mg/L
into molarity (mol/L).
A. Convert mg/L to get g/L then multiply by the volume (L).
B Convert mg/L to g/L then divide by molar
mass (g/mol).
C. Convert mg/L to g/L then divide by the volume (L).
D. Convert mg/L to g/L then divide by the
number of moles (mol).
Answers
The correct option that would convert mg/L into molarity (mol/L)- Convert mg/L to get g/L then multiply by the volume (L). so, option (a) is correct.
What is molarity ?
The amount of solute in one mole per liter of solution is known as molarity. For instance, when table salt is dissolved in water, the solute is salt, and the solution is water. 58.44 grams make up one mole of sodium chloride. One molar solution, often known as 1M, is created when 58.44 grams of sodium chloride are dissolved in one liter of water.
What is volume ?
Volume, a three-dimensional quantity, is used to calculate the capacity of a solid shape. It suggests that the volume of a closed figure determines the amount of three-dimensional space it can occupy.
Therefore, the correct option that would convert mg/L into molarity (mol/L)- Convert mg/L to get g/L then multiply by the volume (L). so, option (a) is correct.
Learn more about molarity from the given link.
https://brainly.com/question/26873446
#SPJ1
Answer: Not A, its B
Explanation: B
A student planned to make copper sulfate crystals from excess copper oxide and dilute sulfuric acid.
The equation for the reaction is:
CuO(s) + H,SO (aq) -, CuSO (aq) + H20(1)
This is the method used.
1. Add 25 cm° of dilute sulfuric acid to a conical flask.
2. Gently warm the dilute sulfuric acid.
3. Add excess copper oxide to the dilute sulfuric acid.
4. Stir the mixture.
5. Heat to evaporate all the water from the mixture.
Suggest two improvements to the method
Explain why each improvement is needed.
A student plans a method to prepare pure crystals of copper sulfate.
The student's method is:
1. Add one spatula of calcium carbonate to dilute hydrochloric acid in a beaker.
2. When the fizzing stops, heat the solution with a Bunsen burner until all the liquid is gone.
The method contains several errors and does not produce copper sulfate crystals.
Explain the improvements the student should make to the method so that pure crystals of copper sulfate are produced.

Answers
The student's method for preparing pure crystals of copper sulfate contains errors and does not produce the desired outcome.
Use copper oxide instead of calcium carbonate: The student should add copper oxide (CuO) to the hydrochloric acid instead of calcium carbonate. Copper oxide reacts with hydrochloric acid to form copper chloride, which can then be converted to copper sulfate through a subsequent reaction with sulfuric acid.
Add sulfuric acid to the copper chloride solution: After the copper chloride solution is formed, the student should add sulfuric acid to it. This reaction between copper chloride and sulfuric acid will yield copper sulfate and hydrochloric acid. The student should ensure that the correct stoichiometric ratio is maintained to maximize the yield of copper sulfate crystals.
Crystal formation: The student should allow the solution to cool slowly after the reaction with sulfuric acid. This promotes the formation of larger, well-defined copper sulfate crystals.
Filtration and drying: Once the crystals have formed, the student should filter the solution to separate the solid crystals from the remaining liquid. The filtered crystals should then be thoroughly dried to remove any remaining water, resulting in pure copper sulfate crystals.
By following these improvements, the student can obtain pure crystals of copper sulfate.
For more such questions on copper sulfate visit:
https://brainly.com/question/17439051
#SPJ8
When is water in a living part of the environment and when is it in a non-living part?
Answers
Answer:
Down >3
Explanation:
The nonliving processes such as evaporation, precipitation, etc... water returns back into the atmosphere.Explanation:Water moves through the help of living organisms in an ecosystem. Although, the non-livings processes are also the main partners of this process.Plants absorbs water through their root system and loss by transpiration. Watere also moves through the living organisms in an ecosystem. Other nonliving processes such as evaporation, precipitation, water returns back into the atmosphere!
Sorry For Miss Spelling >3
A steel tank contains a mixture of Ar and He gases. If the partial pressure of helium in the tank is 1085 mmHg, and the partial pressure of argon is 4867 mmHg, what is the total pressure in the tank (in atm)? Answer as a whole number
Answers
Answer:
8 atm
Explanation:
Given data:
Partial pressure of Ar = 4867 mmHg
Partial pressure of He = 1085 mmHg
Total pressure = ?
Solution:
According to Dalton law of partial pressure,
The total pressure inside container is equal to the sum of partial pressures of individual gases present in container.
Mathematical expression:
P(total) = P₁ + P₂ + P₃+ ............+Pₙ
Now we will solve this problem by using this law.
P(total) = P(Ar) + P(He)
P(total) = 4867 mmHg + 1085 mmHg
P(total) = 5952 mmHg
mmHg to atm;
5952 /760 = 7.83 atm
P(total) = 8 atm
Based on a Kc value of 0.250 and the given data table, what are the equilibrium concentrations of XY, X, and Y , respectively?
Answers
From the solution that we have in the question;
The concentration of X and Y is 0.28 MThe concentration of XY is 0.32 MWhat is the equilibrium constant?The equilibrium constant, denoted as K, is a value that quantitatively represents the ratio of the concentrations of products to reactants at equilibrium in a chemical reaction.
It is a fundamental concept in chemical equilibrium.
The value of the equilibrium constant provides valuable information about the position of equilibrium and the relative concentrations of species involved in a chemical reaction.
Kc = [X] [Y]/[XY]
\(0.25 = (0.1 + x)^2/(0.5 - x)\)
\(0.25(0.5 - x) = (0.1 + x)^2\)
\(0.125 - 0.25x =0.01 + 0.2x + x^2\\ x^2 + 0.45x - 0.115 = 0\)
x = 0.18 M
The equilibrium amount of X and Y= 0.28 M and the equilibrium concentration of XY = 0.32 M
Learn more about equilibrium constant:
https://brainly.com/question/29253884
#SPJ1
Based on the answer to the question that we have;
A 0.28 M concentration of X and Y exists at equilibriumXY's concentration at equilibrium is 0.32 M.The equilibrium constantThe ratio of the product to reactant concentrations in a chemical reaction at equilibrium is represented quantitatively by the equilibrium constant, abbreviated as K.
It is a cornerstone of the theory of chemical equilibrium.
A chemical reaction's equilibrium position and the relative concentrations of the species involved can both be learned from the equilibrium constant's value.
Kc = [X][Y]/[XY]
\(0.25 = (0.1 + x)^2/(0.5 - x)\\0.25(0.5 - x) = (0.1 +x)^2\\0.125 - 0.25x = 0.01 +0.2x +x^2\\= 0.18 M\)
The equilibrium concentration of;
XY =0.5 - 0.18
=0.32 M
Then the equilibrium amount of
X and Y is
0.1 + 0.18= 0.28 M.
Learn more about equilibrium:brainly.com/question/29253884
#SPJ1

Which of the following phase changes involves the transfer of heat from the surroundings to the system?
A
CH4(g)→CH4(l)CH4(g)→CH4(l), because CH4CH4 molecules in the gas phase must absorb energy in order to move closer together, thereby increasing the intermolecular attractions in the solid state.
B
CO2(g)→CO2(s)CO2(g)→CO2(s), because CO2CO2 molecules in the gas phase must absorb energy in order to move closer together, thereby increasing the intermolecular attractions in the liquid state.
C
H2O(l)→H2O(s)H2O(l)→H2O(s), because H2OH2O molecules in the liquid phase must absorb energy in order to create a crystalline structure with strong intermolecular attractions in the solid state.
D
NH3(l)→NH3(g)NH3(l)→NH3(g), because NH3NH3 molecules in the liquid phase must absorb energy in order to overcome their intermolecular attractions and become free gas molecules.
Answers
Because NH3 molecules in the liquid phase require energy to overcome their intermolecular interactions and transform into free gas molecules, this expression reads NH3(l)— NH3(g). Heat is transferred from the environment to the system during phase shifts.
Ammonia has the chemical formula NH3 and is a colourless gas.Nitrogen and hydrogen make up its composition. Its name in aqueous form is ammonium hydroxide. This inorganic substance smells strongly. It is hazardous and corrosive in its concentrated form. Ammonia has a density of 0.769 kg/m3 at STP, making it lighter than air. It is frequently employed as fertiliser. Additionally, it is employed in the production of explosives like TNT and nitrocelluloseAdditionally, it is employed in the manufacture of soda ash and the Ostwald process to produce nitric acid. A strong alkali, such as sodium hydroxide or calcium hydroxide 2NH4Cl + Ca(OH)2 → CaCl2 + 2H2O + 2NH3(g) is heated with an ammonium salt, such as ammonium chloride NH4Cl, to produce ammonia (g)
learn more about ammonium hydroxide Refer: brainly.com/question/12216996
#SPJ4
the atomic mass of the isotope copper-__ is 62.930 amu
Answers
We have that The isotope of copper with the atomic mass of the isotope copper 62.930 amu is Cu 63
Cu 63
From the question we are told
the atomic mass of the isotope copper-__ is 62.930 amu
Generally
Copper is the 29th element of the the periodic table with an atomic mass of 29.
Copper as an elementt has 2 stable isotopes which are
Cu 63
Cu 65
Therefore
The isotope of copper with the atomic mass of the isotope copper 62.930 amu is Cu 63
For more information on this visit
https://brainly.com/question/17756498
If you have 2.60 X 1023 molecules of (NH4)3PO4, how many grams do you have?
Answers
If you have 2.60 x 10²³ molecules of (NH₄)₃PO₄, you have 64.7 grams of it.
The number of particles contained in a sample is measured in terms of the mole. One mole of a compound is the quantity of that substance that has a mass in grams equal to its relative atomic or molecular mass (atomic weight).To find the number of moles of (NH₄)₃PO₄, we'll need to use the Avogadro constant, which is 6.02 x 10²³. We can use the formula:moles = particles ÷ Avogadro constantThe number of particles is given as 2.60 x 10²³. Substituting the values:moles = 2.60 x 10²³ ÷ 6.02 x 10²³moles = 0.432Molar massNow that we have the number of moles of (NH₄)₃PO₄, we can compute its mass. The molecular mass of (NH₄)₃PO₄ is 149.0 g/mol. We can use the formula:mass = moles x molecular mass Substituting the values:mass = 0.432 x 149.0mass = 64.7 grams
For more such questions on molecules
https://brainly.com/question/475709
#SPJ8
Read the sentence. Selena, Ben, and Raj all won leads in the musical, and they has practiced every afternoon during study hall for the past two weeks. Which is the best replacement for the underlined words? have practice will practice will be practicing have practiced
Answers
Answer:
have practiced
Explanation:
Selena, Ben, and Raj all won leads in the musical, and they have practiced every afternoon during study hall for the past two weeks.
Answer:
D.) have practiced
Explanation:
In a chemical reaction, a catalyst changes the ______.
Select one:
a.
heat of reaction
b.
activation energy of the reaction
c.
potential energy of the products
d.
potential energy of the reactants
Answers
Answer:
The Correct answer is B
activation energy of the reaction
or
speeds up the reaction
The average resting heart rate is between
A. 80-90 bpm
B. 90-100 bpm
C. 60-70 bpm
D. 70-75 bpm
Answers
Hope it helps
The average resting heart rate is between is 70- 75 bytes per minute. This include systolic pressure and diastolic pressure. Hence, option D is correct.
What is heart rate?The count of heart beats per unit time is called heart rate. For a normal condition at the resting time the average heart rate is 70 -75 beats per minute.
The heat beats we hear is the impulse from the open and closing of the heart valves. This includes the systolic and diastolic blood pressures. For a normal person at the systolic rate is 120 mm Hg and diastolic rate is 80 mm Hg.
The blood pressure rate is depends on the diet, inheritance and other factors such as age, physical activities, mental situations etc. For a normal person, the average resting heart rate is 70 - 75 bpm.
To find more on heart rate, refer here:
https://brainly.com/question/1155838
#SPJ2
What is a synonym for smart or intelligent cause i cant use either
Answers
Answer:
sharp clever bright
Explanation:
s
Answer:
will you friend me please
I want answer for this question and explaination

Answers
The information that will be the most useful to define a constraint for the device is option D. the temperature at which a reaction becomes unsafe for people
What is the chemical reaction about?The temperature at which a reaction becomes unsafe for people is the most important information to define a constraint for the device, as safety is a primary concern when designing any device for human use. If the reaction releases heat that exceeds a safe temperature, it could cause injury to the user, so it's essential to have a constraint in place to ensure that the reaction stays within safe limits.
Therefore, Other factors such as the average size of a person's foot, the boiling and freezing points of water, and the most popular color for similar products, while important for other aspects of the design, are not as critical for ensuring the safety of the device and its users.
Learn more about chemical reaction at:
https://brainly.com/question/25769000
#SPJ1
See transcribed text below
A chemist is designing a warming device for keeping a person's feet warm when they are outside during cold weather. The device will contain substances that undergo a chemical reaction and release heat.
Which information will be the most useful to define a constraint for the device?
A. the average size of a person's foot
B. the boiling and freezing points of water
C. the most popular color for similar products
D. the temperature at which a reaction becomes unsafe for people
Pls awnser asap!!!
If there is one carbon atom at the beginning of the reaction, how many carbon atoms should be present at the end of the reaction?
A) 0
B) 1
C) 2
D) 4

Answers
Answer:
B) 1
Explanation:
However many you have in the beginning of the reaction, that's how much you have to have at the end of the reaction.
Answer:
B
Explanation:
ye its just B
1. Target I1
Which list of elements all have the same number of valence electrons?
a. F, CI, O, N
b. Cr, Mn, Fe, Co
C. Ne, Ar, Kr, Xe
d. Na, Mg, Al, Si
Answers
Answer:
A
B that is Cr,MN,Fe,Co. is the correct answer
C
D
i have a picture of question

Answers
The mass of the cereal that you would need to consume can be obtained as 84.8 g
What is the number of moles?The number of moles, denoted by the symbol "n," is a fundamental unit of measurement in chemistry that represents the amount of a substance. It is used to quantify the quantity of atoms, molecules, or ions in a sample.
Number of moles of sucrose in the cereal = 11g/342 g/mol
= 0.03 moles
Now;
0.03 moles of sucrose is contained in 60 g of cereal
0.0424 moles of sucrose would contain 0.0424 moles * 60 g/0.03 moles
= 84.8 g
Learn more about moles:https://brainly.com/question/15209553
#SPJ1
Select the correct terms to complete this statement about charged particles.
Like charges attract | repel, and opposite charges attract repel. According to Coulomb's law, as the distance between two charged particles decreases, the force between the particles decreases I increases. As the magnitude of the charges decreases, the force decreases | increases.
Answers
Like charges repel each other, while opposite charges attract each other. This principle is one of the fundamental aspects of electrostatics. According to Coulomb's law, the force between two charged particles is directly proportional to the product of their charges and inversely proportional to the square of the distance between them.
As the distance between two charged particles decreases, the force between them increases. This is because the closer the particles are, the stronger the electric field they create, leading to a stronger force of interaction.
On the other hand, as the magnitude of the charges decreases, the force between the particles also decreases. This is because the force is directly proportional to the product of the charges. If one or both of the charges are smaller, the force they exert on each other will be weaker.
In summary, according to Coulomb's law, decreasing the distance between charged particles increases the force between them, while decreasing the magnitude of the charges decreases the force. This understanding of the relationship between charge, distance, and force is crucial in explaining the behavior of charged particles and the interactions between them.
Know more about Coulomb's law here:
https://brainly.com/question/26892767
#SPJ8
Write the balanced equation for the equilibrium reaction for the dissociation ofsilver chloride in water, and write the K expression for this reaction. Then create an ICE chart. Since we know the equilibrium concentration of the silver ion, we can solve for Ksp.Does it agree with the literature value
Answers
Answer:
See explanation
Explanation:
Hello there!
In this case, since the the concentrations are not given, and not even the Ksp, we can solve this problem by setting up the chemical equation, the equilibrium constant expression and the ICE table only:
\(AgCl(s)\rightleftharpoons Ag^+(aq)+Cl^-(aq)\)
Next, the equilibrium expression according to the produced aqueous species as the solid silver chloride is not involved in there:
\(Ksp=[Ag^+][Cl^-]\)
And therefore, the ICE table, in which x stands for the molar solubility of the silver chloride:
\(\ \ \ \ \ \ \ \ \ \ \ \ \ \ AgCl(s)\rightleftharpoons Ag^+(aq)+Cl^-(aq)\)
I - 0 0
C - +x +x
E - x x
Which leads to the following modified equilibrium expression:
\(Ksp=x^2\)
Unfortunately, values were not given, and they cannot be arbitrarily assigned or assumed.
Regards!
The decomposition of hydrogen peroxide follows first order reaction kinetics with a rate constant k = 6.40 x 10-3 s-1.
2 H2O2(aq) ⟶ 2 H2O(l) + O2(g)
If the reaction starts with an H2O2 concentration of 6.80 M, how long will it take for the H2O2 concentration to reach 2 M?
Answers
In this first-order the decomposition reaction, the H2O2 concentration will drop from 6.80 M to 2 M in roughly 191.1 seconds.
The first-order rate equation can be used to calculate how long it will take for the hydrogen peroxide (H2O2) concentration to reach 2 M:
ln([H2O2]t/[H2O2]0) = -kt
Where:
The amount of H2O2 present at time t is known as [H2O2]t.H2O2 is initially present at a concentration of zero, and k is the rate constant.
To find t, we can rewrite the equation as follows:
t = -[ln([H2O2]t/[H2O2]0)] / k
Plugging in the given values:
[H2O2]t = 2 M
[H2O2]0 = 6.80 M
\(k = 6.40 \times 10^{(-3)} s^{(-1)\)
t = -[ln(2/6.80)] / (6.40 x 10^(-3))
Now, we can calculate the time:
t ≈ -[ln(0.2941)] / (6.40 x 10^(-3))
t ≈ -(−1.2231) / (6.40 x 10^(-3))
t ≈ 191.1 seconds
for more quesions on decomposition reaction
https://brainly.com/question/14608831
#SPJ8
What is true about dominant alleles? a They almost never appear as the trait. b They appear as the trait only when there are two of them c They appear as the trait over a recessive allele d They appear as the trait if there is not recessive allele
Answers
They appear as the trait over a recessive allele. Statement C) is true about the dominant alleles.
Dominant alleles are genetic variants that, when present in an individual's genotype, are expressed phenotypically, meaning they determine the visible or observable traits. Dominant alleles are represented by capital letters, while recessive alleles are represented by lowercase letters in genetics.
In terms of inheritance, if an individual has at least one copy of the dominant allele, it will be expressed in the phenotype, regardless of the presence of a recessive allele. This is because dominant alleles exert their influence over recessive alleles, thus "dominating" their expression.
To illustrate this, let's consider a specific example using a trait controlled by a single gene with two possible alleles: dominant (A) and recessive (a). If an individual is homozygous dominant (AA), meaning they possess two copies of the dominant allele, the dominant trait will be expressed.
However, if an individual is homozygous recessive (aa), with two copies of the recessive allele, the recessive trait will be expressed since there are no dominant alleles to override it.
Therefore, dominant alleles appear as the trait over recessive alleles, regardless of the presence or absence of a recessive allele. The presence of even a single copy of the dominant allele is sufficient for its expression in the phenotype. Option C
For more such questions on recessive allele visit:
https://brainly.com/question/16048928
#SPJ8
PLS HELP ASAP.thank you so much if u help me!!!

Answers
which is the graph of the function g(x) = f(-x)
Answers
To graph the function g(x) = f(-x), you can start with the graph of f(x) and then reflect it about the y-axis.
What is a graph of the function g(x) = f(-x)?To find the graph of the function g(x) = f(-x), we can start with the graph of the function f(x) and then reflect it about the y-axis.
If the graph of f(x) is symmetric with respect to the y-axis, meaning it is unchanged when reflected, then g(x) = f(-x) will have the same graph as f(x).
However, if the graph of f(x) is not symmetric with respect to the y-axis, then g(x) = f(-x) will be a reflection of f(x) about the y-axis.
In either case, the resulting graph of g(x) = f(-x) will be symmetric with respect to the y-axis.
Learn more about the graph of functions at: https://brainly.com/question/17089414
#SPJ1

Which of the following can be mixed in solution with NH3 to make a buffer?
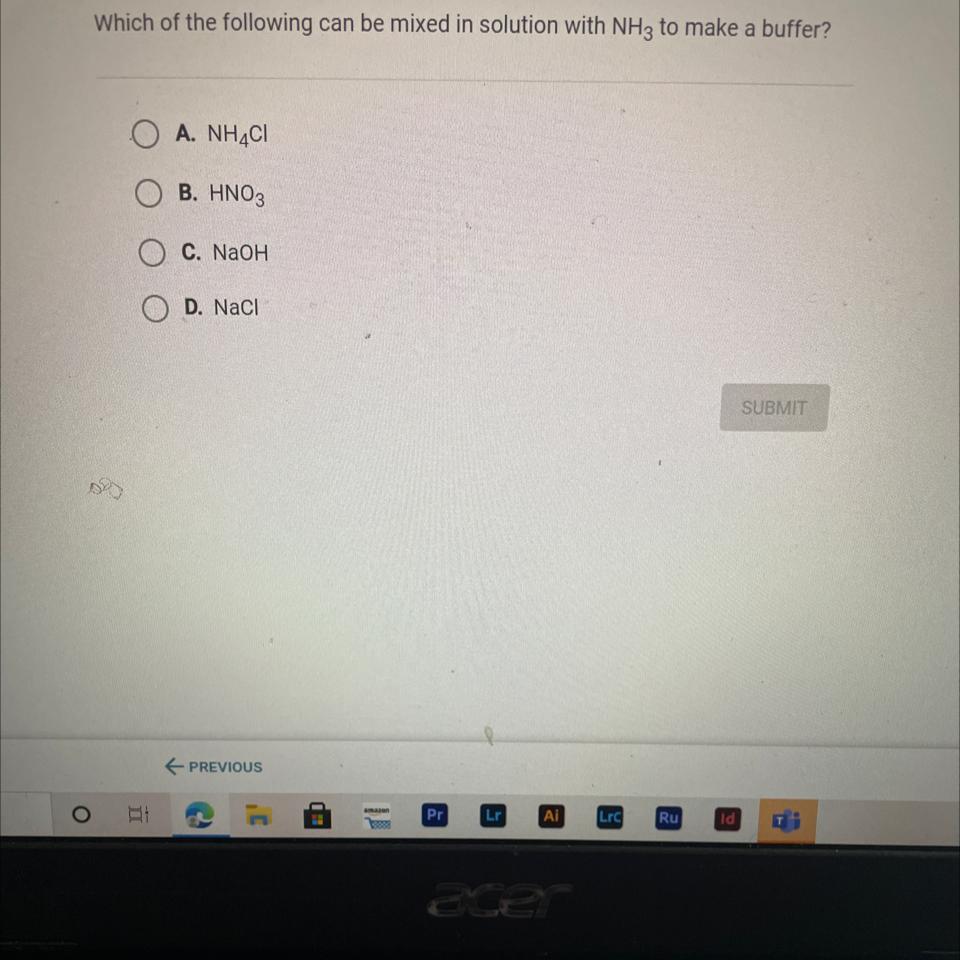
Answers
Answer:
c. NaOH
Explanation:
just took the quiz